Your Gut Microbiome: From In Utero to Older Life
The gut microbiome plays a foundational role in human health across the lifespan. From early development in utero to the ageing process in older adults, shifts in microbial diversity and composition impact immunity, metabolism, and risk of disease.
Prenatal Beginnings: A Sterile Womb or Not?
For over a century, researchers have debated how the fetal gut is first colonised. Two key theories exist: the “in utero colonisation” hypothesis and the “sterile womb” paradigm.(1-4) The former suggests that microbial exposure begins before birth, while the latter proposes that the prenatal environment is sterile. Conflicting evidence supports both views.
Microbial Development in Early Life: The First 1,000 Days
While the exact timing of first colonisation is debated, most microbial exposure happens during and after birth. The transition from a relatively sterile womb to a microbe-rich environment marks a critical window for gut and immune development.(4) Factors such as delivery mode, antibiotics, and feeding practices strongly shape the infant microbiome.(5)
The first 1,000 days are only part of the microbiome’s journey.
This visual gives useful context for how the gut microbiome continues to adapt throughout life. Let’s now take a closer look at the foundations established in infancy.
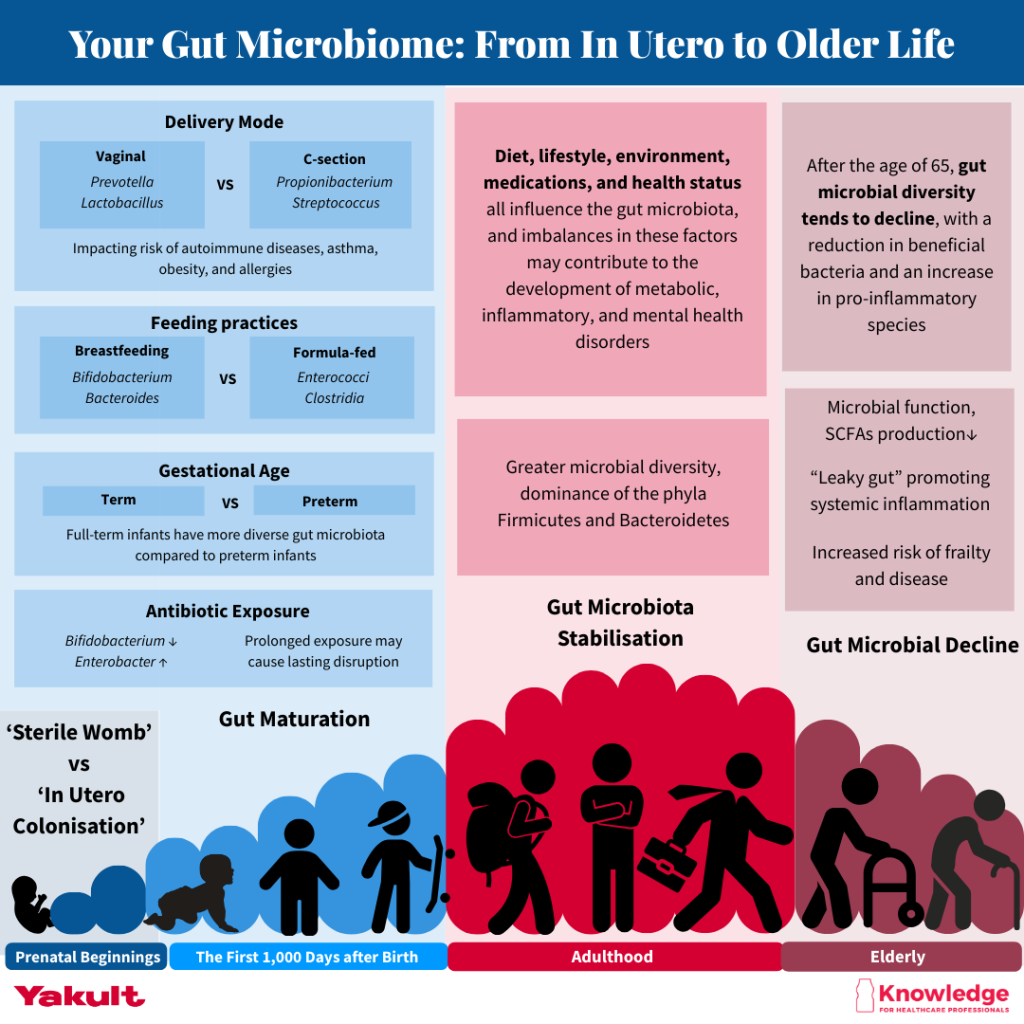
Delivery mode
Gut microbiota composition differs markedly depending on birth method.(7–9) Vaginally delivered infants are colonised by maternal vaginal and gut microbes like Prevotella and Lactobacillus.(6,7) Caesarean-born infants instead acquire skin-associated microbes like Propionibacterium and Streptococcus,(8,9) and often carry more antibiotic-resistance genes. (2)
Epidemiological studies have linked caesarean section birth to an increased risk of autoimmune diseases, asthma, obesity, and allergies.(10) These differences in infant microbiota between vaginal and C-section deliveries are often attributed to the so-called “bacterial baptism”, where vaginal microbes are vertically transmitted to the newborn to support microbial colonisation.(11)
However, alternative explanations have been proposed. Some researchers argue that differences in infant microbiota following C-section may be influenced more by factors such as intrapartum antibiotic use, maternal inactivity, or complications during pregnancy, rather than the absence of vaginal exposure alone.(11) The current studies may overestimate the effect of vaginal delivery on infant microbiota.(12) Therefore, convincing evidence is necessary to clarify the differences in infant gut microbiota caused by the mode of delivery.
Gestational age at birth:
Full-term infants tend to develop a more diverse gut microbiota that matures more rapidly toward an adult-like composition compared to preterm infants.(12,13) Premature babies face unique challenges, such as prolonged hospitalisation, frequent antibiotic use, and parenteral nutrition, all of which delay or disrupt microbiome development.(14,15)
Antibiotic exposure:
Antibiotics are often necessary in the early days of life, particularly for preterm infants at risk of infection. However, evidence shows that even short courses can temporarily reduce gut microbial diversity, especially lowering beneficial Bifidobacterium levels and allowing potentially harmful bacteria like Enterobacter to dominate.(16,17) Prolonged exposure may cause more lasting disruption, potentially increasing the risk of conditions like necrotising enterocolitis (NEC).(18-20) While antibiotics can be life-saving, they also disrupt the delicate balance of the developing microbiota.(21)
Probiotics, particularly strains such as Lactobacillus rhamnosus GG and Bifidobacterium spp have been shown to reduce the risk of NEC and support microbiota restoration, though strain selection and safety must be carefully considered in vulnerable infants.(22)
Feeding practices: Breast-fed vs formula-fed
Human milk oligosaccharides (HMOs) in breastmilk act as prebiotics that nourish beneficial gut microbes like Bifidobacterium, Lactobacillus, and Bacteroides. These microbes break down HMOs into smaller sugars that also support other bacteria, promoting a balanced microbiome and improved nutrient utilisation.(23,24) Consequently, breastfed and formula-fed infants develop distinct gut microbiota profiles.
Over the past two decades, research has shown that Bifidobacteria are the dominant microbes in the guts of breastfed infants, while formula-fed infants tend to have microbiota dominated by Enterococci and Clostridia.(25) Interestingly, a study by Bezirtzoglou et al.(26) reported that while formula-fed infants had a lower total bacterial load, they exhibited greater species diversity compared to their breastfed counterparts, highlighting the compositional and functional differences driven by early feeding practices.
According to the World Health Organization (WHO) and UNICEF, breastfeeding should begin within the first hour after birth and continue exclusively for the first six months, meaning no other food or drink, not even water.(27) Infants should be fed on demand, day and night, and without the use of bottles, teats, or pacifiers. From six months onward, safe, age-appropriate complementary foods should be introduced while continuing breastfeeding up to two years of age or beyond.
Beyond its immediate nutritional value, breastfeeding offers long-term health benefits. Breastfed children tend to achieve higher scores on intelligence tests and are at lower risk of becoming overweight, obese, or developing type 2 diabetes later in life. For mothers, breastfeeding has been associated with a reduced risk of breast and ovarian cancers.(27)
Introduction to solid foods and maturation of infant gut
The functions of the gut microbiota shift notably before and after the introduction of solid foods. During the first year of life, the early microbiota is dominated by bacteria that primarily utilise lactate.(14) As solid foods are introduced, the microbial community expands to include bacteria capable of metabolising a broader range of carbohydrates, producing vitamins, and breaking down xenobiotics.(2,14)
Additionally, the introduction of non-digestible carbohydrates during weaning supports the maturation of pancreatic function, small intestinal absorption, and colonic fermentation in infants. These changes alter the overall environment of the digestive tract and influence the types of substrates that reach the developing colon.(28)
Overall, introducing solid foods appears to trigger the maturation of the infant gut microbiota, gradually shifting it toward an adult-like composition. However, further research is needed to identify which specific components of the solid food diet most strongly influence microbiota development, and how different feeding patterns impact overall infant health.

Adulthood to Older Age: Stability, Shifts, and Health Implications
By the age of three, the gut microbiota typically stabilises into an adult-like configuration, characterised by greater microbial diversity and a dominance of the phyla Firmicutes and Bacteroidetes.(29) Throughout adulthood, the microbiota remains relatively stable under healthy conditions, although it can be influenced by diet, lifestyle, environment, medications (especially antibiotics), and health status.
A fibre-rich, diverse diet continues to be a major determinant of microbiota health, supporting the production of short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate. These SCFAs serve as key modulators of gut barrier integrity, inflammation, and energy metabolism, and mental health.(30)
Emerging evidence suggests SCFAs influence brain function via the gut-brain axis, affecting neurotransmitter production, stress response, and behaviour, with potential links to mood disorders such as anxiety and depression.(30) Conversely, high-fat, low-fibre Western-style diets have been associated with dysbiosis, reduced microbial diversity, and increased risk of metabolic and inflammatory diseases.(31)
What Changes After 65?
After the age of 65, the gut microbiome typically shows reduced diversity and a decline in beneficial bacteria like Bifidobacterium and Faecalibacterium prausnitzii, alongside an increase in pro-inflammatory microbes such as Enterobacteriaceae. This imbalance contributes to chronic low-grade inflammation, or “inflammaging”.(32,33)
Ageing is also linked to a decline in microbial function, notably reduced production of SCFAs, which may impact frailty, cognition, and gut barrier integrity. Increased gut permeability, or “leaky gut,” allows bacterial components like lipopolysaccharides (LPS) to enter the bloodstream, promoting systemic inflammation and raising the risk of conditions such as cardiovascular disease, neurodegeneration, and autoimmunity.(34) Microbial imbalances may also disrupt the gut-brain axis, affecting neurotransmitter production and contributing to cognitive decline and mood disorders in older adults.(34)
Interestingly, some anti-aging effects of commonly used drugs may also be mediated by the microbiota. For example, metformin, a first-line treatment for metabolic disease, has been shown to exert pro-longevity effects by influencing the gut microbiota, particularly through the suppression of bacterial folate metabolism.(34) This highlights the emerging role of microbial metabolites in modulating health and longevity.
A systematic review suggests that probiotics may offer some benefits for older adults, particularly in areas like muscle strength, physical function, and potentially cognitive function.(35) However, the evidence is not conclusive, and more robust research is needed, especially with larger, well-defined populations, and focusing on specific outcomes.
Take home message:
- The gut microbiome is a dynamic ecosystem that evolves throughout life. It is shaped by early exposures, diet, medications, and physiological changes associated with ageing. From immunity development in infancy to inflammation control in older age, the microbiome is central to lifelong health.
- Understanding these microbial shifts opens the door to targeted strategies, like optimising early feeding practices, limiting unnecessary antibiotic exposure, and incorporating fibre-rich, diverse diets that support gut health from birth through the ageing process.
- For vulnerable populations such as preterm infants or the elderly, evidence-based use of probiotics may also offer therapeutic benefits in reducing disease risk and maintaining microbial balance.
- As research continues to uncover the complex interplay between microbes and human health, nurturing a resilient gut microbiota remains a cornerstone of preventive health across the lifespan.
References:
- Perez-Muñoz ME, Arrieta MC, Ramer-Tait AE. et al. A critical assessment of the “sterile womb” and “in utero colonization” hypotheses: implications for research on the pioneer infant microbiome. Microbiome. 2017;5;48. doi: https://doi.org/10.1186/s40168-017-0268-4
- Stinson LF, Boyce MC, Payne MS, Keelan JA. The Not-so-Sterile Womb: Evidence That the Human Fetus Is Exposed to Bacteria Prior to Birth. Front. Microbiol. 2019; 10. doi: https://doi.org/10.3389/fmicb.2019.01124
- Banchi P, Colitti B, Opsomer G, Rota A, Van Soom A. The dogma of the sterile uterus revisited: does microbial seeding occur during fetal life in humans and animals? Reproduction. 2023 13;167(1):e230078. doi: 10.1530/REP-23-0078. PMID: 37903182; PMCID: PMC10762539.
- Panzer JJ, Romero R, Greenberg JM, Winters JG, Lopez NG, Thies KR. Is there a placental microbiota? A critical review and re-analysis of published placental microbiota datasets. BMC Microbiol. 2023;23: 76. doi: https://doi.org/10.1186/s12866-023-02764-6
- Robertson RC, Manges AR, Finlay BB, Prendergast AJ. The Human Microbiome and Child Growth – First 1000 Days and Beyond. Trends Microbiol. 2019; 27(2): 131-137. https://www.cell.com/trends/microbiology/fulltext/S0966-842X(18)30204-X?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS0966842X1830204X%3Fshowall%3Dtrue
- GMFH Edititng Team. Discover how your gut microbiota evolves during your lifespan with this new Infographic, Gut Microbiota for Health. 2019; Accessed 21st July 2025. Available at: https://www.gutmicrobiotaforhealth.com/discover-how-your-gut-microbiota-evolves-during-your-lifespan-with-this-new-infographic/
- Dominguez-Bello, M. G., Costello, E. K., Contreras, M., Magris, M., Hidalgo, G., Fierer, N., et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. U.S.A. 2010:107:11971–11975. doi: 10.1073/pnas.1002601107
- Jakobsson, H. E., Abrahamsson, T. R., Jenmalm, M. C., Harris, K., Quince, C., Jernberg, C., et al. Decreased gut microbiota diversity, delayed Bacteroidetes colonisation and reduced Th1 responses in infants delivered by caesarean section. Gut 63. 2014:559–566. doi: 10.1136/gutjnl-2012-303249
- Backhed, F., Roswall, J., Peng, Y., Feng, Q., Jia, H., Kovatcheva-Datchary, P., et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe. 2015; 17: 690–703. doi: 10.1016/j.chom.2015.05.012
- Stokholm J, Thorsen J, Blaser MJ, Rasmussen MA, Hjelmso M, Shah S, et al. Delivery Mode and Gut Microbial Changes Correlate With an Increased Risk of Childhood Asthma. Sci Transl Med. 2020;12(569):eaax9929. doi: 10.1126/scitranslmed.aax9929
- Stinson LF, Payne MS, Keelan JA. A Critical Review of the Bacterial Baptism Hypothesis and the Impact of Cesarean Delivery on the Infant Microbiome. Front Med (Lausanne). 2018;5:135. doi: 10.3389/fmed.2018.00135
- Yao Y., Cai X., Ye Y., Wang F., Chen F., Zheng C. The Role of Microbiota in Infant Health: From Early Life to Adulthood. Front. Immunol. 2021;12:708472. doi: 10.3389/fimmu.2021.708472
- Arboleya S., Binetti A., Salazar N., Fernández N., Solís G., Hernandez-Barranco A., Margolles A., de los Reyes-Gavilán C.G., Gueimonde M. Establishment and development of intestinal microbiota in preterm neonates. FEMS Microbiol. Ecol. 2012;79:763–772. doi: 10.1111/j.1574-6941.2011.01261.x
- Pantazi AC, Balasa AL, Mihai CM, Chisnoiu T, Lupu VV, Kassim MAK, Mihai L, Frecus CE, Chirila SI, Lupu A, Andrusca A, Ionescu C, Cuzic V, Cambrea SC. Development of Gut Microbiota in the First 1000 Days after Birth and Potential Interventions. Nutrients. 2023;15(16):3647. doi: 10.3390/nu15163647.
- Tirone C, Pezza L, Paladini A, Tana M, Aurilia C, Lio A, D’Ippolito S, Tersigni C, Posteraro B, Sanguinetti M, et al. Gut and Lung Microbiota in Preterm Infants: Immunological Modulation and Implication in Neonatal Outcomes. Front. Immunol. 2019;10:2910. doi: 10.3389/fimmu.2019.02910.
- Tanaka S, Kobayashi T, Songjinda P, Tateyama A, Tsubouchi M, Kiyohara C, Shirakawa T, Sonomoto K, Nakayama J. Influence of antibiotic exposure in the early postnatal period on the development of intestinal microbiota. FEMS Immunol. Med. Microbiol. 2009;56:80–87. doi: 10.1111/j.1574-695X.2009.00553.x
- Fouhy F, Guinane CM, Hussey S, Wall R, Ryan CA, Dempsey EM, Murphy B, Ross RP, Fitzgerald GF, Stanton C, et al. High-Throughput Sequencing Reveals the Incomplete, Short-Term Recovery of Infant Gut Microbiota following Parenteral Antibiotic Treatment with Ampicillin and Gentamicin. Antimicrob. Agents Chemother. 2012;56:5811–5820. doi: 10.1128/AAC.00789-12.
- Greenwood C, Morrow AL, Lagomarcino AJ, Altaye M, Taft DH, Yu Z, Newburg DS, Ward DV, Schibler KR. Early Empiric Antibiotic Use in Preterm Infants Is Associated with Lower Bacterial Diversity and Higher Relative Abundance of Enterobacter. J. Pediatr. 2014;165:23–29. doi: 10.1016/j.jpeds.2014.01.010.
- Morreale C, Giaroni C, Baj A, Folgori L, Barcellini L, Dhami A, Agosti M, Bresesti I. Effects of Perinatal Antibiotic Exposure and Neonatal Gut Microbiota. Antibiotics (Basel). 2023;12(2):258. doi: 10.3390/antibiotics12020258.
- Cordero L, Ayers LW. Duration of empiric antibiotics for suspected early-onset sepsis in extremely low birth weight infants. Infect Control Hosp Epidemiol 2003;24:662-6. doi: 10.1086/502270.
- Deshpande G, Rao S, Patole S, Bulsara M. Updated meta-analysis of probiotics for preventing necrotizing enterocolitis in preterm neonates. Pediatrics 2010;125:921-30. doi: 10.1542/peds.2009-1301
- Akker C, Goudoever JB, Shamir R, Domellof M, Embleton ND, Hojsak I, et al. Probiotics and Preterm Infants. J Pediatr Gastroenterol Nutr. 2020; 70 (5): 664-680. doi: https://doi.org/10.1097/MPG.0000000000002655
- Beck, LC, Masi, AC, Young, GR. et al. Strain-specific impacts of probiotics are a significant driver of gut microbiome development in very preterm infants. Nat Microbiol. 2022;7:1525–1535. https://doi.org/10.1038/s41564-022-01213-w
- Davis EC, Castagna VP, Sela DA, Hillard MA, Lindberg S, Mantis NJ, Seppo AE, Järvinen KM. Gut microbiome and breast-feeding: Implications for early immune development. J Allergy Clin Immunol. 2022;150(3):523-534. doi: 10.1016/j.jaci.2022.07.014.
- Balmer, S. E., and Wharton, B. A. (1989). Diet and faecal flora in the newborn: breast milk and infant formula. Arch. Dis. Child. 64, 1672–1677. doi: 10.1136/adc.64.12.1672
- Bezirtzoglou, E., Tsiotsias, A., and Welling, G. W. (2011). Microbiota profile in feces of breast- and formula-fed newborns by using fluorescence in situ hybridization (FISH). Anaerobe 17, 478–482. doi: 10.1016/j.anaerobe.2011.03.009
- Davis EC, Castagna VP, Sela DA, Hillard MA, Lindberg S, Mantis NJ, Seppo AE, Järvinen KM. Gut microbiome and breast-feeding: Implications for early immune development. J Allergy Clin Immunol. 2022;150(3):523-534. doi: 10.1016/j.jaci.2022.07.014. PMID: 36075638; PMCID: PMC9463492.
- Parrett AM, Edwards CA. In vitro fermentation of carbohydrate by breast fed and formula fed infants. Arch Dis Child. 1997;76:249–53. doi:10.1136/adc.76.3.249
- Shreiner AB, Kao JY, Young VB. The gut microbiome in health and in disease. Curr Opin Gastroenterol. 2015;31(1):69-75. doi: 10.1097/MOG.0000000000000139.
- Silva YP, Bernardi A, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020; 11 – 2020. Doi: https://doi.org/10.3389/fendo.2020.00025
- Severino A, Tohumcu E, Tamai L, Dargenio P, Porcacri S, Rondinella D. The microbiome-driven impact of western diet in the development of noncommunicable chronic disorders. Best Pract Res Clin Gastroenterol. 2024; 72 – 101923. doi: https://doi.org/10.1016/j.bpg.2024.101923
- Parsaei M, Sarafraz N, Moaddad SY, Leylabadlo HE. The importance of Faecalibacterium prausnitzii in human health and diseases. New Microbes New Infect. 2021; 43:100928. doi: 10.1016/j.nmni.2021.100928.
- Ragonnaud E, Biragyn. Gut microbiota as the key controllers of “healthy” aging of elderly people. Immunity & Ageing. 2021; 18(2). Doi: https://doi.org/10.1186/s12979-020-00213-w
- Nagpal R, Mainali R, Ahmadi S, Wang S, Singh R, Kavanagh K, Kitzman DW, Kushugulova A, Marotta F, Yadav H. Gut microbiome and aging: Physiological and mechanistic insights. Nutr Healthy Aging. 2018;4(4):267-285. doi: 10.3233/NHA-170030.
- Handajani YS, Turana Y, Hengky A, Hamid G, Schroeder-Butterfill E, Kristian K. Probiotics supplementation or probiotic-fortified products on sarcopenic indices in older adults: systematic review and meta-analysis from recent randomized controlled trials. Front Aging. 2024;5:1307762. doi: 10.3389/fragi.2024.1307762.
